Analysis of Organometallics
Unlike other techniques, LIFDI is able to characterize difficult samples. The protocol is easy and quick. Information is available immediately after the synthesis, confirming the molecular weight if the reaction succeeded.
LIFDI can be used complementary to traditional methods like X-Ray to increase the throughput. It can deliver data even in cases where X-Ray, NMR and elemental analysis fail. See J. Am. Soc. Mass Spectrom., 2013 characterizing an
- oily, not crystallizable mixture of air sensitive cyclic tetrastibane homologues
- with scrambling propyl and ethyl ligands decomposing upon chromatography
LIFDI is much faster than X-Ray. The entire analysis including sample preparation under inert conditions is performed within minutes.
LIFDI gives intense molecular ion signals verifying the measured isotope pattern with the theoretical pattern.
Example: Cp*2UCl2
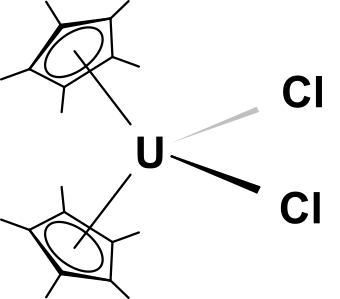
“The M[η5-(CH3)5C5]2R2 and M[η5-(CH3)5C5]2R(Cl) complexes of thorium and uranium are the most reactive and chemically versatile actinide hydrocarbyls studied to date.” Synthesis & quote according to: J. Am. Chem. Soc., 1981
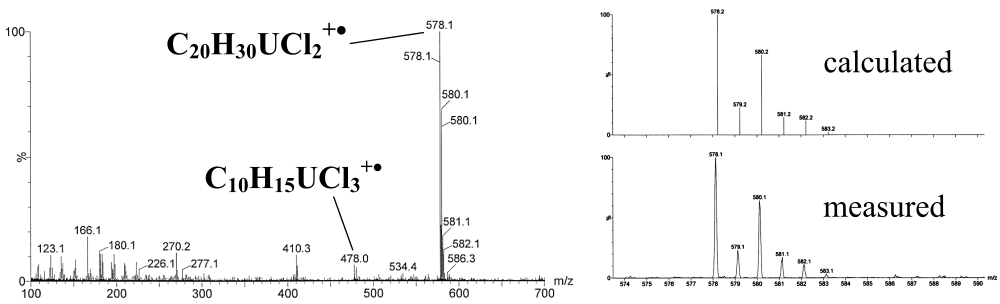
LIFDI was able to ionize the intact molecule and shows a perfect match between the calculated and measured isotope pattern.
Structural information
While LIFDI usually provides the intact molecular ion, fragmentation can be induced by collision or thermally to provide structural information.
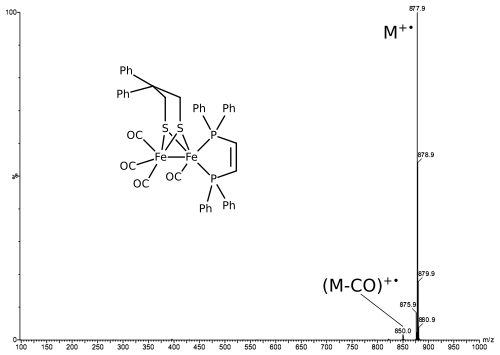
The spectrum shows the intact molecular ion according to Chem. Eur. J., 2013
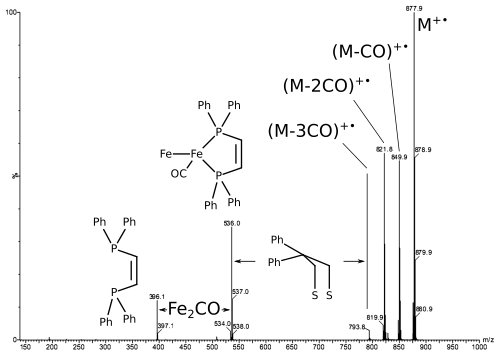
Thermally induced fragmentation support the structure of the molecule.
Reactive & Air Sensitive
A LIFDI analysis can easily be performed under inert conditions.

Reactive samples are synthesized in a glove box and bottled in a septum capped vial as usual. The headspace contains inert gas at slight overpressure.
For analysis, the LIFDI-capillary is pierced through the septum and dipped into the sample solution for 2-3 seconds. Ca. 40nL are aspirated and travel through the capillary to the emitter in the ion source. This way the sample is always protected by inert gas.
Example: Grubbs’ Catalyst

Grubbs’ Catalyst is a Ruthenium-Carbene-complex used for Olefin metathesis reactions, discovered by Robert H. Grubbs. Grubbs’ Catalyst is air sensitive and thus handling needs extra care. However the LIFDI-analysis is quick and easy and the LIFDI Mass Spectrum shows the true molecular ion with high intensity.
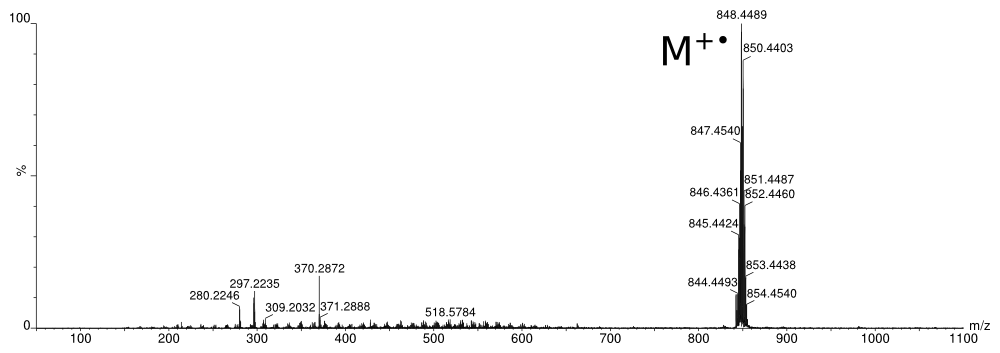
The zoom shows the pronounced Ruthenium isotope-pattern matches the calculated one nicely.
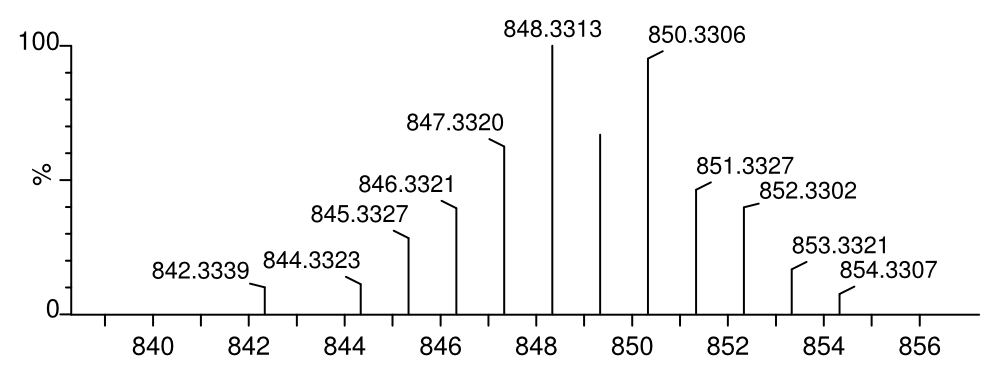
calculated:
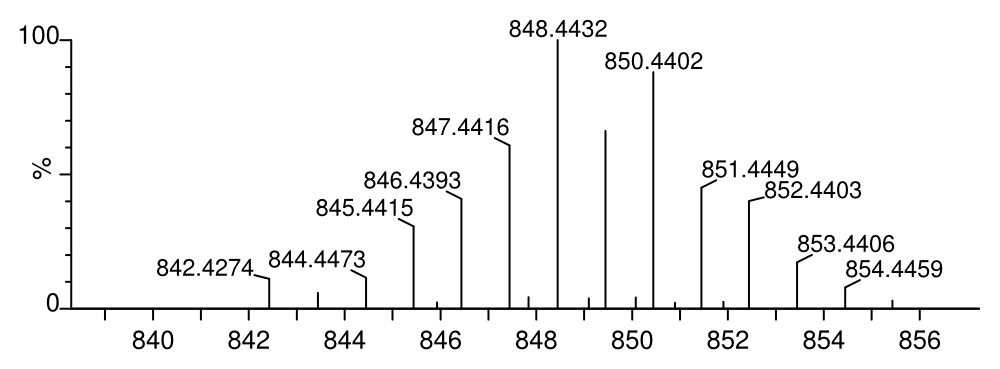
measured:
Even for difficult samples, this analysis can be conducted a few minutes after the sample has been prepared in the glove box.
Example: chromium phenyl hydride
C64H88N4Cr2 [{({iPr2(C6H3)}2nacnac)Cr}2(m-Ph)(m-H)]
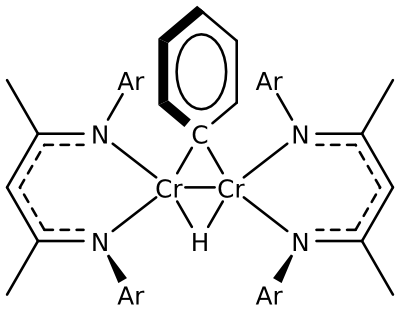
The extremely air-sensitive di-chromium complex was synthesised at the university of Delaware.
ESI, EI and other ionization methods didn’t exhibit the molecular ion. X-Ray revealed the structure, but the presence of the hydride ligand bridging the two chromium atoms had to be proved by H/D exchange and a shift of the molecular mass by 1 Da.
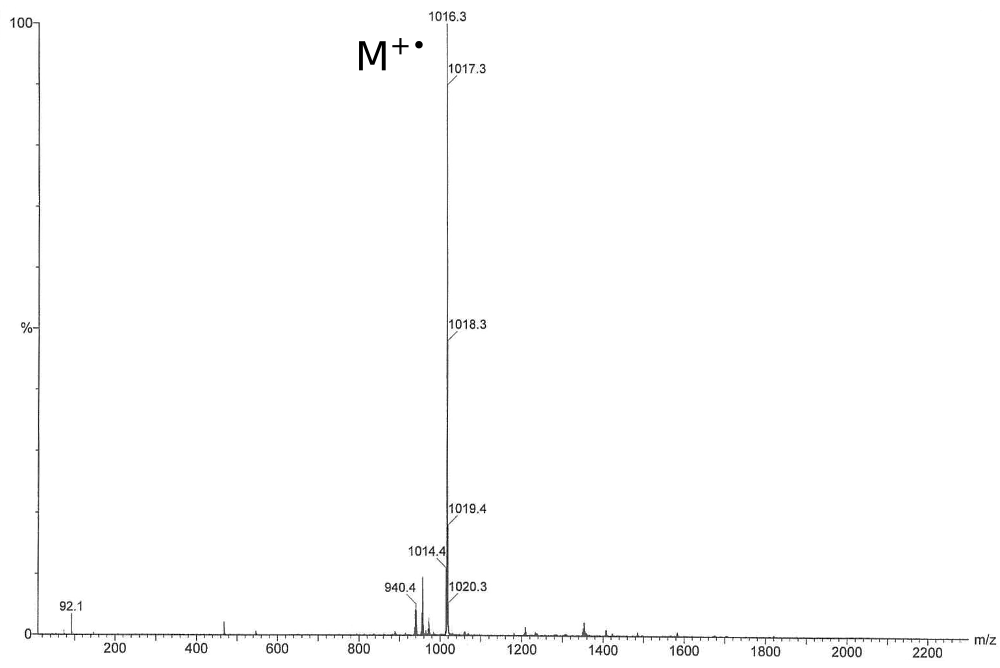
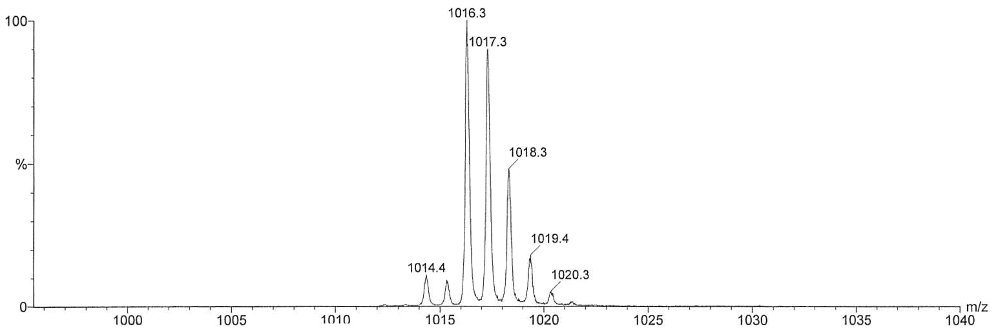
This spectrum shows the undeuterated compound. See the publication for more information: Angew. Chem. Int. Ed., 2007
calculated isotope pattern
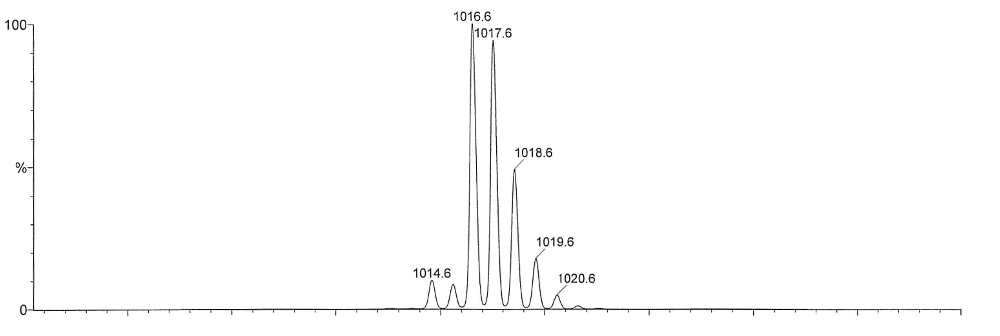
measured isotope pattern
Literature
More examples can be found in the literature, for instance:
Liquid Injection Field Desorption Ionization Mass Spectrometry of Cyclic Metal Carbonyl Complexes with Tetra-Antimony Ligands J. Am. Soc. Mass Spectrom., 2013 24, 164-166Measured on JEOL AccuTOF with LIFDI
Liquid injection field desorption/ionization of transitional metal fluoride complexes J. Fluorine Chem., 2010 1213-1217Measured on Waters GCT-Premier with LIFDI
Liquid injection field desorption/ionization of reactive transition metal complexes Anal. Bioanal. Chem., 2006 386, 52-58Measured on JEOL JMS 700 with LIFDI
Liquid injection field desorption ionization: a new tool for soft ionization of samples including air sensitive catalysts and non-polar hydrocarbons Eur. J. Mass Spectrom., 2004 10, 459 - 468Measured on several instruments with LIFDI
